What if the next medicine against drug resistant infections did not come from a factory on Earth, but from a biology experiment floating in orbit? That idea just moved a step closer to reality.
A new study finds that viruses evolved in space can be redesigned into powerful tools against bacteria that no longer respond to common antibiotics.
The work, led by researchers at University of Wisconsin–Madison with partners at Rhodium Scientific, appears in the open access journal PLOS Biology. It tackles a crisis that health agencies now rank among the biggest threats to public health.
Bacterial antimicrobial resistance was directly responsible for about 1.27 million deaths in 2019 and linked to nearly 5 million in total. Urinary tract infections are one of the most common reasons doctors prescribe antibiotics in clinics and emergency rooms, which means they play a big part in this problem.
Space as an evolutionary test kitchen
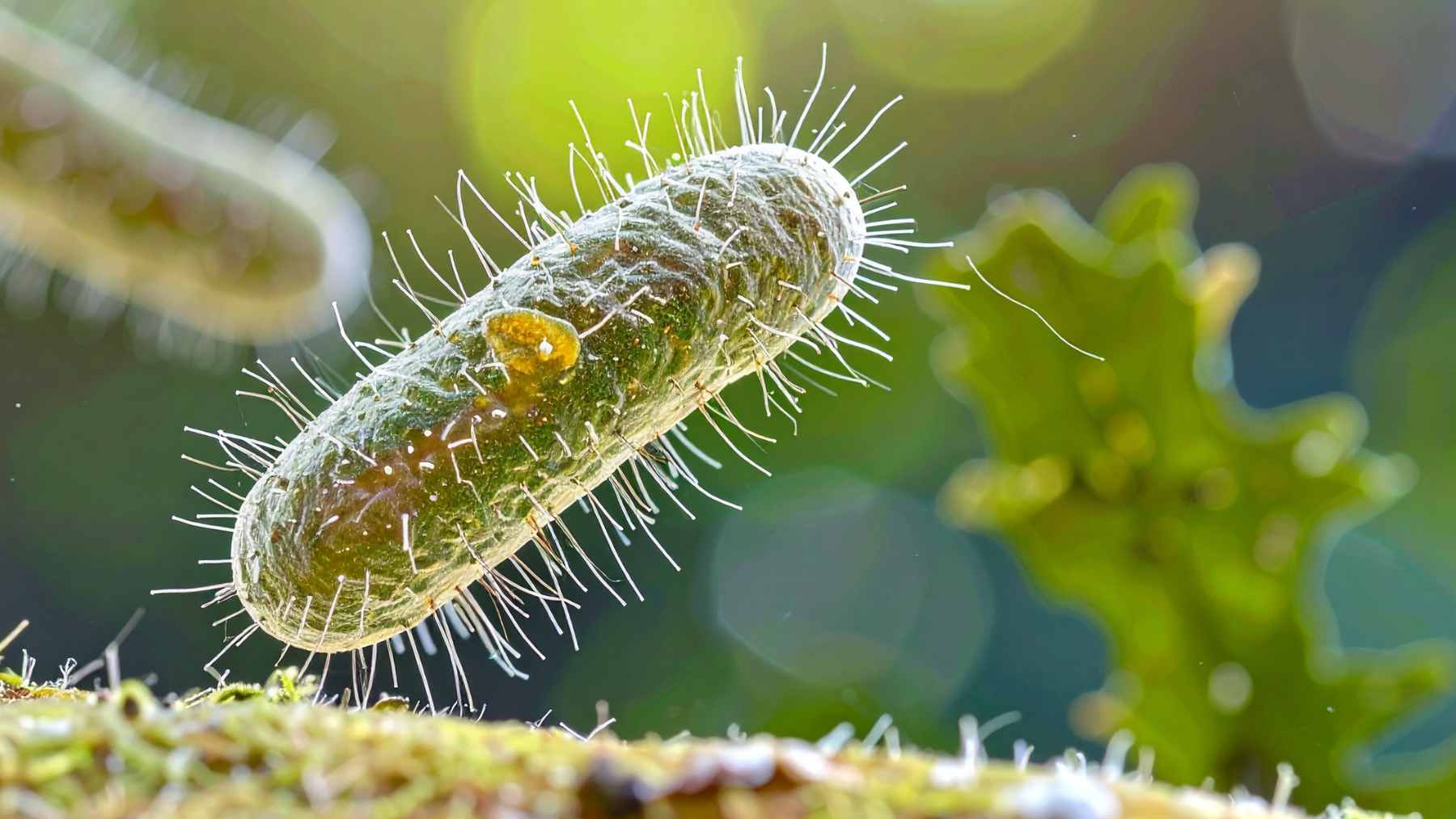
To explore how near weightlessness changes the battle between viruses and bacteria, the team packed bacteria called Escherichia coli together with a virus named T7 into special frozen vials.
These flew to the International Space Station while identical samples stayed on Earth as controls. In orbit, scientists incubated the mixtures for periods from a few hours up to 23 days, then refroze everything and sent it back to the lab.
At first, the space viruses struggled. On Earth, T7 typically wipes out its bacterial host within hours. In microgravity, infection started much more slowly, likely because there is almost no natural stirring of the liquid.
Phages and bacteria had to bump into each other mainly by random motion, which is a bit like trying to find a friend in a crowded room where no one is moving.
Under that stress, the microbes began to change. Bacteria evolved mutations in a gene called mlaA that usually helps keep fatty molecules tucked inside the outer membrane. In space, some bacteria flipped more of those molecules outward.
That altered the surface where viruses normally latch on. In response, the most successful phages evolved new changes in the protein tip they use to grab bacteria, including hydrophobic substitutions that likely made the viral tail fibers more flexible or more stable on those strange surfaces.
From space mutants to UTI killers
Back on Earth, the team took the mutations that worked best in space and combined them into redesigned T7 viruses.
When they tested these against uropathogenic E. coli that cause urinary tract infections, the space-informed phages were able to kill strains that resisted the original virus. In contrast, similar phage variants evolved only under Earth gravity showed no extra activity against these tough pathogens.
That matters for everyday life. UTIs are a familiar reason for a prescription, a trip to the pharmacy, and a week of pills on the nightstand. Yet more than 90% of the bacteria that cause these infections already resist at least one antibiotic.
If new phage therapies can take pressure off traditional drugs, doctors could use fewer broad-spectrum antibiotics and slow the spread of resistance.
A new role for space in sustainable medicine
Scientists are careful to stress that this is early stage research. The engineered phages were tested in the lab, not yet in patients, and any future treatments would need strict safety checks as well as ways to manufacture them at scale.
Still, the study hints at a future where orbiting labs act as evolutionary “accelerators,” helping researchers discover virus designs that are hard to find under normal gravity.
In practical terms, that could mean more targeted therapies that spare our microbiomes, reduce unnecessary antibiotic use, and support healthier ecosystems of microbes in people and in hospitals. It is a small step, but it points to a surprising ally in the fight against superbugs: the quiet microgravity inside a spacecraft.
The study was published in PLOS Biology.