On modern Mars, dust storms are doing far more than painting the sky orange. A new laboratory study finds that the Red Planet’s constant haze of dust is powering electrical reactions that turn ordinary salts into toxic chemicals in the air and soil, effectively turning the atmosphere into what some researchers call a world that is “actively manufacturing poison.”
For future astronauts dreaming about a Mars base, that is not just an abstract chemistry problem. It is something that could end up in air filters, drinking water, and even the dust that sticks to a spacesuit after a long walk outside.
Strange isotopes, strange planet
The puzzle started with a mismatch in the numbers. Orbiters and rovers have repeatedly measured the ratios of light and heavy versions of elements like chlorine, oxygen, and carbon on the Martian surface and in its thin air.
Heavy isotopes, especially chlorine 37, are much less common than expected, by about fifty parts per thousand for chlorine and smaller but still significant amounts for oxygen and carbon.
On Earth, big isotope shifts usually point to big processes such as boiling oceans, long-lived lakes, or deep burial in rocks. Mars does not have those today. So where was the missing heavy chlorine going, and why did so many samples from different missions tell the same story?
Dust storms as giant chemical reactors
The new work, led by Neil Sturchio of the University of Delaware and Alian Wang of Washington University in St. Louis, recreates Martian dust storms inside specialized vacuum chambers.
Under low pressure carbon dioxide, mineral grains grind past one another until they build up static electricity, much like rubbing a balloon on your hair or feeling a tiny shock when you touch a metal doorknob on a dry winter day.
Once the charge is high enough, small electrical arcs jump through the gas. Those arcs knock energetic electrons into carbon dioxide molecules and create very reactive fragments, including carbon monoxide and atomic oxygen.
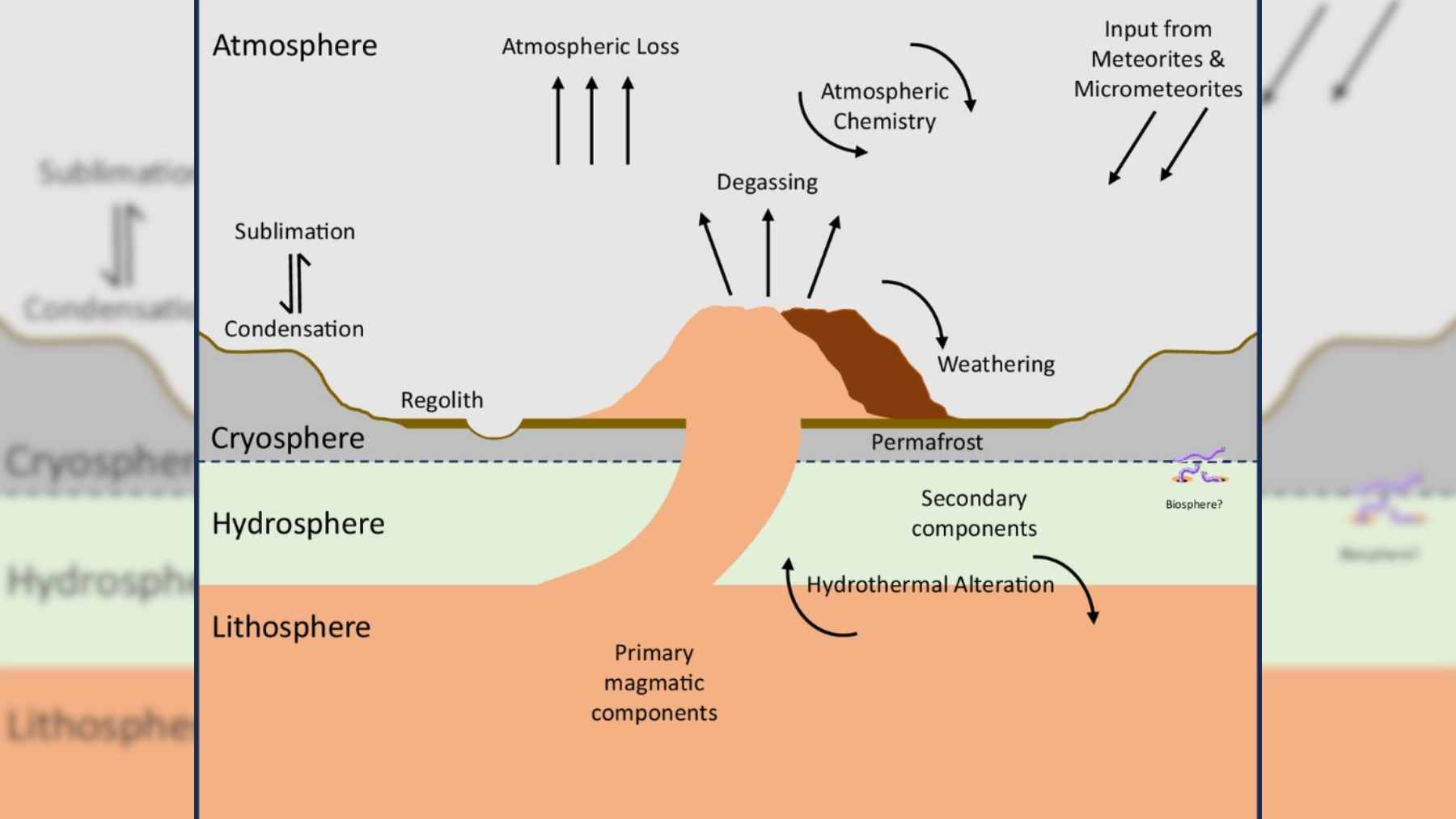
In the experiments, those fragments then attack chloride salts in simulated Martian soil and convert them into more oxidized compounds such as chlorates and perchlorates, along with airborne carbonates and other oxidants.
Over time, this electrical chemistry selectively uses lighter isotopes of chlorine, oxygen, and carbon more readily than heavy ones.
The result is a surface and atmosphere that are systematically depleted in heavy isotopes, matching the odd values seen by missions like Curiosity and European orbiters much better than earlier models that relied on liquid water and heat.
What this means for human explorers
Perchlorates are not just a curiosity in a chemistry textbook. They are highly reactive oxidants and they interfere with the human thyroid, which regulates metabolism.
Studies suggest that Martian soils hold perchlorates in the range of about 0.5 to 1 percent by weight, based on measurements from the Phoenix lander and later work, a level considered toxic for long-term human exposure.
On a dusty world where fine particles can stay suspended for weeks, that matters. Every gust that rattles a habitat wall or scours a solar panel could loft small amounts of perchlorate-rich dust into the air around a future base. Filtering it out of breathing systems, wash water, and even greenhouse soils would become part of routine life, not a one time engineering challenge.
At the same time, some researchers point out that perchlorates can be useful. They could serve as ingredients for rocket oxidizers or even help form stronger bricks, turning a hazard into a resource if handled with care.
The new study does not remove that possibility, but it does underline how dynamic and persistent the source of these chemicals is on present day Mars.
Rethinking Mars’s past, and its neighbors
The findings also nudge scientists to be more cautious when using minerals such as carbonates as proof of ancient lakes or seas. If electrical discharges in dry dust can build some of those compounds today, then at least part of the mineral record might come from cold, arid chemistry instead of warm, watery environments.
Perseverance has already recorded dozens of electrical discharges inside Martian dust devils, confirming that this is not just a laboratory curiosity but an everyday feature of the planet’s weather. Similar processes may quietly shape the surfaces of other worlds such as Venus or Titan, where charged dust and lightning also have room to work.
For people planning the first long stays on the Red Planet, the message is simple. Mars is not only cold and dry, it is electrically active and chemically busy, constantly refreshing a layer of toxins that explorers will need to manage.
The study was published in Earth and Planetary Science Letters.









