A team in Switzerland has built a microrobot about the size of a grain of sand that can travel through blood vessels, drop off medicine exactly where it is needed, and then dissolve.
The device is guided from outside the body with carefully controlled magnetic fields and has already passed key tests in realistic vessel models and in pigs, which have vascular systems similar to ours.
If that sounds like science fiction, think about what happens when you swallow a painkiller for a headache. The pill does not just go to your forehead. It floods your whole body, which is why so many promising drugs fail during development and why strong treatments can come with brutal side effects.
This microrobot aims to flip that script by turning drug delivery into something much more precise.
How the microrobot moves inside the body
The new device is a tiny, spherical capsule made from a soluble gel. Inside are three key ingredients: iron oxide nanoparticles that let magnets grab and move it, a radiology contrast agent so doctors can see it on X ray images, and the drug itself.
Researchers at ETH Zurich pair the capsule with a custom catheter and a modular electromagnetic navigation system that fits into an operating room. The capsule is first injected into blood or cerebrospinal fluid through the catheter, then the magnetic system takes over.
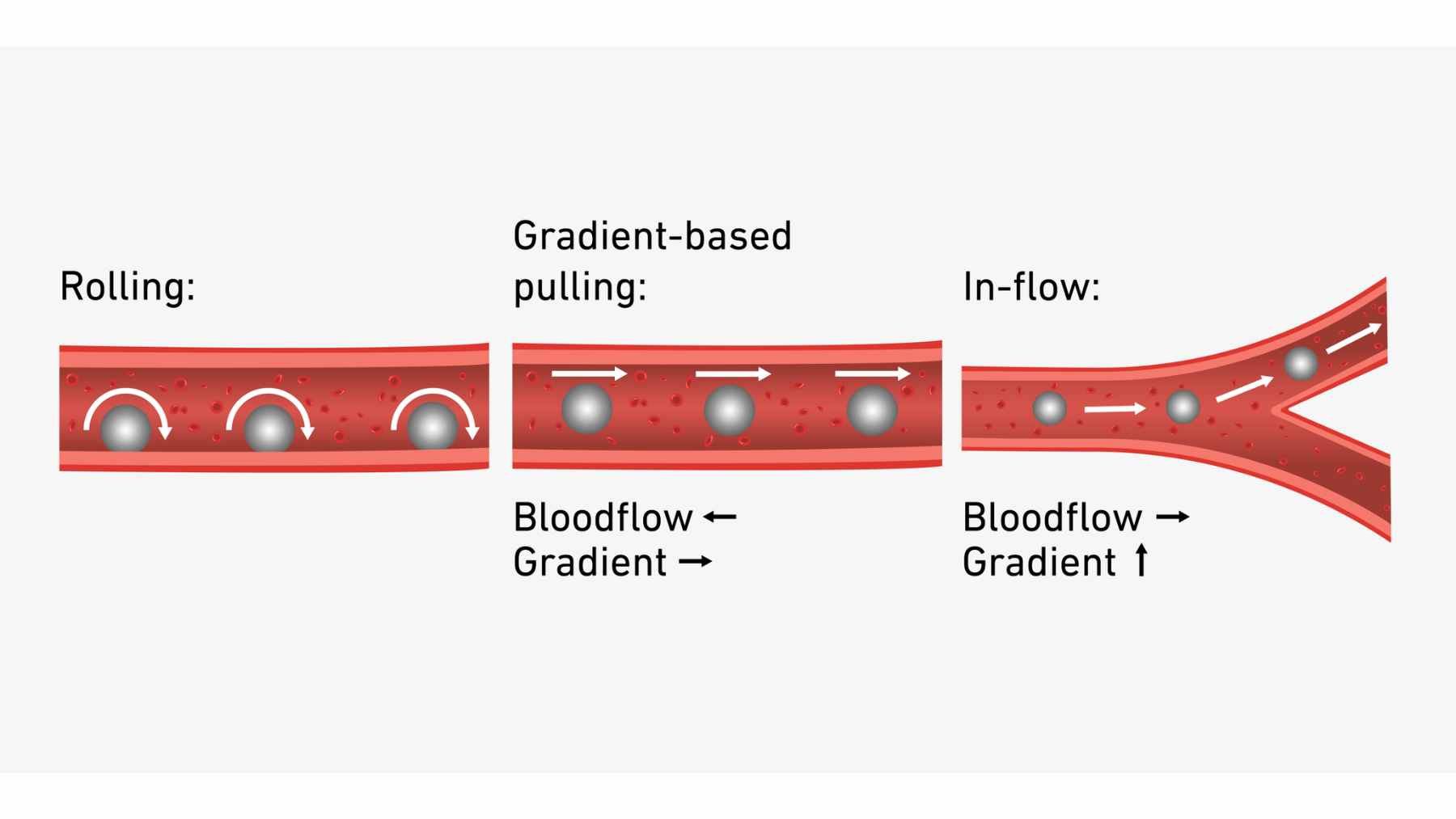
By mixing different magnetic field patterns, the team can roll the capsule along vessel walls or pull it through branching arteries, even when blood is racing past at more than twenty centimeters per second. In tests, they could steer it at about four millimeters per second with very fine control.
Crucially for doctors watching the monitors, the capsule shows up clearly on fluoroscopy, a type of live X-ray imaging. Once it reaches the target area, a high-frequency magnetic field gently heats the nanoparticles, melting the gel shell so the drug spills out locally and the capsule itself dissolves.
Early results in animal arteries
Before going anywhere near human patients, the team rehearsed in painstaking detail. They first used silicone models of human and animal blood vessels that are realistic enough to serve as training tools for surgeons. In these models, the microrobot could be guided through twisting paths to a blood clot and release a clot busting drug directly onto it.
Only after those dry runs did they test in pigs and in the cerebrospinal fluid of a sheep. Under fluoroscopy, the capsule stayed visible from start to finish and reached its intended destination in more than 95% of cases, a striking success rate for something this small in fast flowing blood.
Why this matters for stroke and cancer care
Every year, about twelve million people worldwide suffer a stroke, often caused by a blood clot that blocks vessels feeding the brain. Current clot dissolving drugs are delivered through the bloodstream and spread everywhere, so doctors must use relatively high doses that can trigger dangerous bleeding elsewhere in the body.
By sending a tiny capsule right to the clot and nowhere else, clinicians could in principle use lower doses and still get a strong local effect. The same logic applies to aggressive brain tumors, aneurysms and abnormal vessel tangles known as arteriovenous malformations, all of which may benefit from therapy placed with pinpoint accuracy instead of flooding the whole body.
Big promise, but still early days
The microrobot described in the new work is not a consumer gadget. It is a complex system that has to balance enough magnetic material to steer, enough contrast agent to see, and enough room left over for a meaningful dose of medicine.
The supporting hardware is not trivial either, from the electromagnetic coils to the catheter design.
Experts who were not involved in the project say the effort marks a turning point for medical robotics. Roboticist Howie Choset described it as by far the most exciting work he has seen for delivering high precision care, while also warning that the field still faces major hurdles as it moves toward real-world use.
For patients and families, the practical question is simple. Could future treatments feel less like carpet bombing the body and more like a careful repair job on just the damaged spot? The answer will depend on how upcoming safety tests and eventual human trials play out, but to a large extent the roadmap is now clearer than it has ever been.
The study was published in Science.









