If you live with aching knees or a back that never really shuts up, you probably know the routine of reaching for painkillers or planning your day around flare ups. Now scientists are testing something very different in the lab.
Instead of numbing pain in the brain, an experimental therapy nicknamed a “pain sponge” tries to stop those signals right where they start.
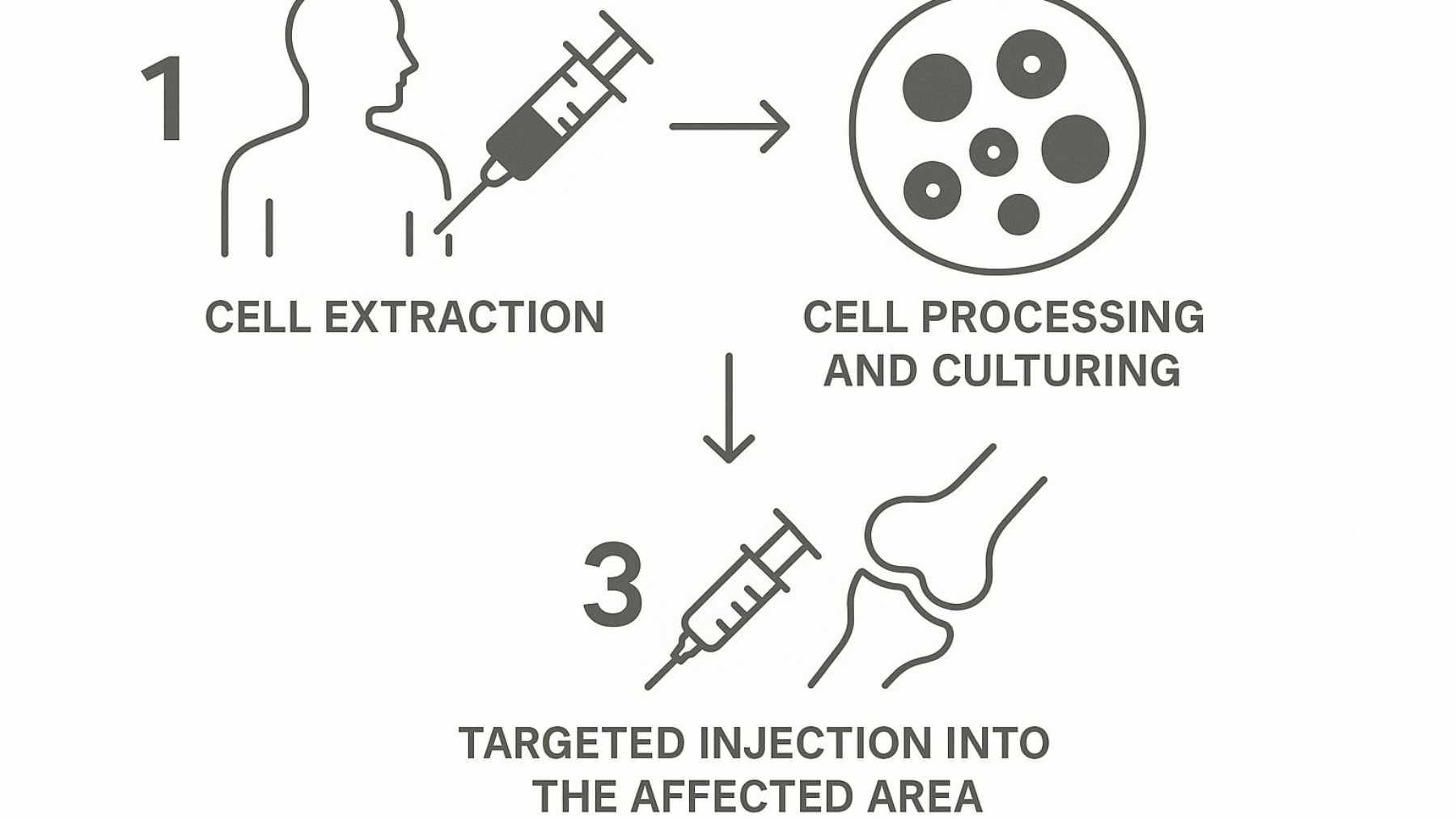
In a new preclinical study, researchers at Johns Hopkins School of Medicine engineered human stem cells into pain sensing neurons and injected them into arthritic mouse knees.
These lab-grown cells acted as biological decoys, soaking up inflammatory signals that would normally trigger chronic pain and, at the same time, helping protect bone and cartilage in the joint.
For people living with long-term pain, that matters. In recent national data, roughly one in four U.S. adults reported chronic pain lasting three months or more, a burden closely tied to lost work days and opioid use. Osteoarthritis, the joint disease targeted in this study, is already a leading cause of disability worldwide.
How the “pain sponge” works
The therapy, called SN101, starts with human pluripotent stem cells that can become many different cell types. Researchers coaxed them into specialized sensory neurons known as nociceptors, the cells that normally detect harmful heat, pressure, or chemical irritation.
Instead of replacing the body’s own neurons, these transplanted cells share the joint space with them. In the mouse experiments, a carefully selected subset of these neurons behaved like a sponge for inflammatory molecules in the knee.
They bound pain triggering factors and kept them from reaching native neurons, which meant fewer pain signals were sent toward the brain. At the same time, protein analyses suggested the grafted cells released factors that supported bone and cartilage repair, so the joint environment itself began to shift in a healthier direction.
“Our approach utilizes high-purity stem cell derived nociceptors that effectively function as a sponge for pain factors,” explained Gabsang Lee, a scientific co-founder of SereNeuro Therapeutics.
A different path from pills and injections
Right now, osteoarthritis pain is usually managed with lifestyle changes, nonsteroidal anti-inflammatory drugs, steroid injections, and sometimes opioids. Those medicines can help, but they often bring stomach problems, cardiovascular risks, or dependence, and they mostly work by dampening how the brain perceives pain rather than calming the storm inside the joint.
By contrast, SN101 aims at the source. In the arthritic mice, animals given these engineered neurons showed less pain related behavior and less joint damage than control animals, hinting at a therapy that may ease pain while slowing structural deterioration.
Orthopedics expert Chuan-Ju Liu, who was not involved in the work, told Live Science that “the possibility that the therapy could both relieve pain and slow cartilage degeneration is particularly compelling for osteoarthritis.”
For patients who rely on opioid prescriptions just to get through the day, the idea that a one time or infrequent joint injection could eventually reduce that need is understandably appealing.
Still early days for the pain sponge
There is an important catch. Everything so far has been done in mice. The underlying research appears as a preprint, and while it has now moved through peer review, the approach has not yet reached human trials.
Researchers still have to show that these cells are safe over the long term, that they do not trigger harmful immune reactions, and that what works in a mouse knee can translate to the much larger and more complex human joint.
Even then, any future treatment would have to pass formal toxicology testing and multiple phases of clinical trials before regulators could consider it.
Still, the work points to a quiet shift in how scientists think about pain. Instead of treating nociceptors as enemies to silence, the SN101 strategy treats certain pain-sensing neurons as tools that can be redeployed to intercept danger signals and help repair damaged tissue.
For people juggling heating pads, physical therapy appointments, and bottles of pills, that is at least a glimpse of a different future.
The study was published in bioRxiv.